Dharshi Devendran, Berkeley Science Review
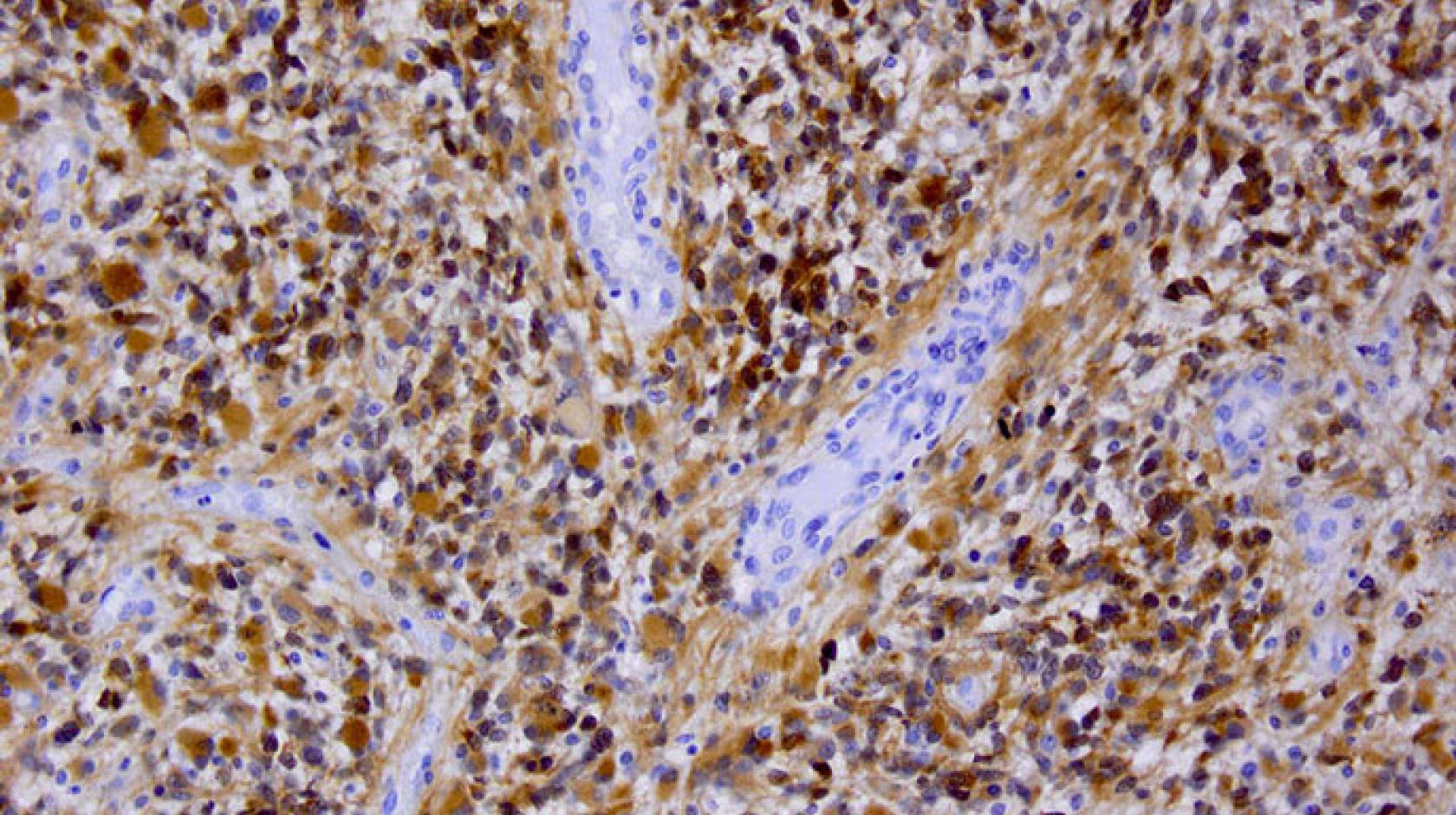
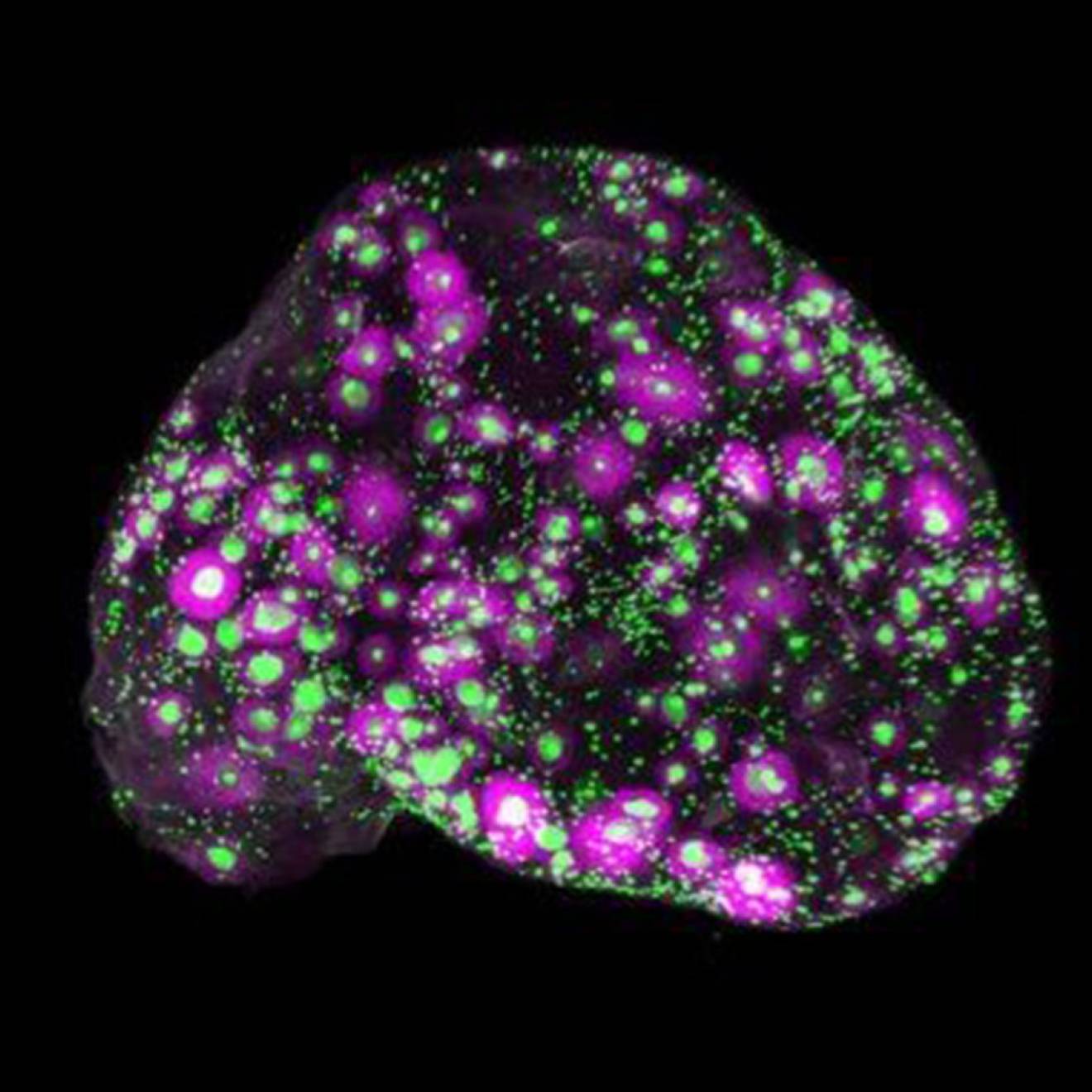
The destruction starts with a modification in the instructions that tell a single brain cell how to grow. As the cell rapidly multiplies into thousands of cancerous cells, an irregularly shaped tumor mass forms, with tendrils that invade deeper into healthy brain tissue. The tumor’s abnormal growth makes it hungry for more oxygen and nutrients, so it hijacks the circulatory system and forces new blood vessels to grow around it. It is resistant to radiation, and is protected by the brain-blood barrier—a series of defenses within blood vessel walls that protect the brain from toxic molecules, but also keep out cancer drugs that could kill the cancerous cells. Left to continue growing, it eventually destroys its host. This is the brain cancer glioblastoma multiforme.
Although it is difficult to fight, glioblastoma also has a weakness. In its rush to feed itself, it accelerates the blood vessel formation process and creates hole-riddled blood vessels around the tumor. Because cancer drugs are small enough to slip through these holes, they can exploit this defect—but they also need a strategy to cross the blood-brain barrier in order to reach the tumor.
To help cancer drugs access the tumor, Ting Xu, professor of materials science at UC Berkeley, and her collaborators are making tiny particles called nanocarriers that protect the drugs during their journey to the tumor. They recently demonstrated that their nanocarrier, called a 3-helix micelle (3HM), is twice as effective at reaching glioblastoma cells as a liposome, a nanocarrier that is currently the gold standard in drug-delivering nanotechnology.
Read the full article at Berkeley Science Review.