Grant Burningham, UCSF

Inspiration can be a hard thing to find.
The history of science is filled with elusive “eureka moments” taking place under unlikely circumstances — Archimedes’ jump in a bath to intuit displacement, Issac Newton’s observation of a falling apple to grasp gravity, and Nikola Tesla’s inspiration for the electric induction motor, which came as he was observing a sunset in a park and quoting Faust. In the halls of UC San Francisco, sometimes inspiration comes knocking on the door.
Shuvo Roy, Ph.D., is a UCSF bioengineer working on the world’s first implantable bioartificial kidney. He also works down the hall from hundreds of people at the top of their scientific fields, in a place where scientists can make serendipitous connections just by walking out of their office doors.
That was how Roy came to partner with an embryonic stem cell expert, surgeons, fellow bioengineers, and diabetes experts, and how his work to create a bioartificial kidney spun off into something totally new — work to create a bioartificial pancreas.
“One of the amazing things about UCSF is the resources you have access to,” Roy said.
A silicon filter
Roy, a faculty member in the Department of Bioengineering and Therapeutic Sciences, a joint department of the UCSF Schools of Pharmacy and Medicine, has been spending the better part of a decade working with colleagues nationwide on what would be a sea change for patients with end-stage kidney disease: a surgically implantable bioartificial kidney. The device removes toxins from the blood through a silicon nanotechnology-based filter with microscopic holes. It also uses kidney cells to perform biological functions of the natural kidney, applying tissue engineering techniques.
Work on the device has been progressing at a steady clip. The earliest human trials are expected to begin in months.
But it was during animal trials that Andrew Posselt, M.D., Ph.D., the director of the Pancreatic Islet Transplant Program in the School of Medicine’s Department of Surgery, suggested to Roy that the technology used in the bioartificial kidney had the potential to be used to create a bioartificial pancreas as well.
Treating diabetes
Credit: Wikimedia Commons
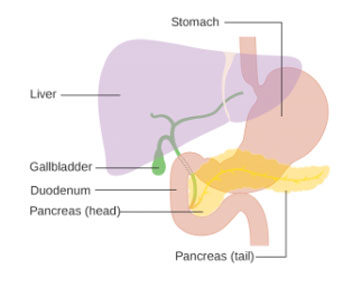
Diabetes is a disease of the pancreas. The small organ tucked under the liver produces the hormone insulin, which regulates blood sugar. When it doesn’t produce enough insulin, blood sugar can spike, causing damage throughout the body.
There is an interesting symmetry between diabetes and the kidney project—diabetes is the leading cause of kidney failure and the reason many people need dialysis to begin with.
Clinicians have long dreamed of surgically implanting insulin-producing cells in diabetes sufferers and end the need for either medication or daily insulin injections. The problem is that the body’s immune system tends to attack the implanted hormone-producing cells and kill them.
There are medications that keep the body from rejecting implants, but over time the anti-rejection drugs also kill the implanted cells.
One solution is to make a device or pouch with holes small enough that the immune system cells can’t get in to kill the insulin-producing cells, but smaller things, like nutrients and oxygen, can.
Semi-permeable pouches are one possible solution, but one hurdle they face is that they work on diffusion — the principle that nutrients, or any other substance, will naturally disperse from high concentrations to low ones, creating an even distribution. Insulin produced in these artificial organs would diffuse out of the pouch like a tea bag sitting in a cup of hot water. Roy is developing a different approach.
Roy’s bioartificial kidney uses pressure-driven filtration, with the heart as the pump. A bioartificial pancreas that works off the same idea could keep the implanted cells alive for longer by forcing nutrients in the device using blood pressure. Roy’s precisely printed holes also have to keep out white blood cells, the body’s immune cells, which are 20-50 micrometers, or about one-thousandth of an inch. And they also have to allow smaller things, like oxygen atoms and sugar, to enter the organ.
In theory, Roy’s technology could be used to create a small, credit card-sized implantable device that could change diabetes treatment.
A team to make it happen

Credit: Steve Babuljak
Building a functioning bioartificial pancreas will require the top minds of multiple scientific disciplines.
The Diabetes Center at UCSF has been a key contributor. But the team also needs bioengineers to design the device, surgeons to figure out how to connect the device to the circulatory system, and experts in the biology of the cells that produce insulin.
Tejal Desai, Ph.D., chair of the Department of Bioengineering and Therapeutic Sciences, has been a key connector.
Desai helped Roy make one of the team’s most vital connections, to Matthias Hebrok, Ph.D., the director of the Diabetes Center at UCSF. Hebrok is an expert on the islets of Langerhans, the cells that actually produce insulin in the pancreas. Hebrok has created these islets from stem cells and knows what it will take to keep them alive.
Surgery is another big piece of the puzzle. Because the bioartificial pancreas needs actual blood flow and must be reliably implanted, Roy is also collaborating with other UCSF Department of Surgery faculty members, including Shant Vartanian, M.D., a vascular surgeon with expertise in making connections between blood vessels and medical devices, and Qizhi Tang, Ph.D., an expert in immune system response to transplants, along with Posselt, who initially suggested the bioartificial pancreas.
“This is an example of how great science and generous collegiality produces more great science,” Roy said, adding that the borderless culture of UCSF allows experts from different disciplines to come together to advance health care. “This is a truly special place,” he said.

Credit: Barbara Ries
The project is in the middle of a three-year National Institute of Health (NIH) grant for $2.4 million and two-year California Institute of Regenerative Medicine (CIRM) grants, totaling $1.1 million to work on a treatment for Type I diabetes.
The spirit of collaboration is part of UCSF’s DNA, so much so that Desai, who founded a company to create a different type of artificial pancreas doesn’t see any competition in helping make another one.
“One of them will work,” she said, adding that several pancreas projects are going on at UCSF. “It’s going to take multiple interactions, approaches, and perspectives to find a solution.”

