Levi Gadye, UC San Francisco
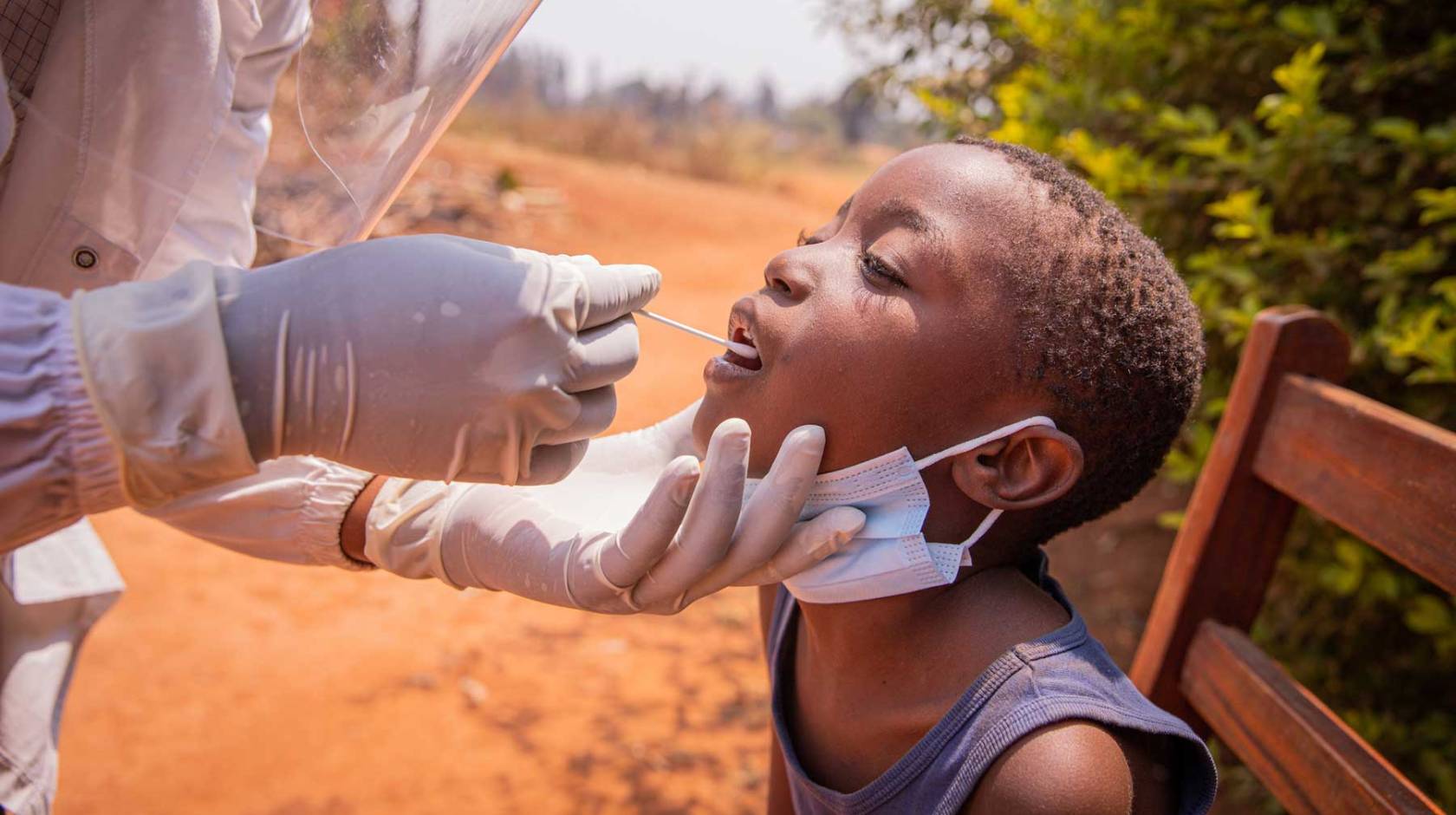
Drugs to treat tuberculosis have been around for more than 75 years, yet it remains the world’s top infectious disease killer. A big obstacle has been testing. It’s either inaccurate — missing up to half of all cases — or requires expensive laboratories that are not widely available in the countries where most people with TB live.
This could change with the arrival of a portable device called MiniDock MTB that can deliver accurate test results in less than half an hour. In a study published April 29 in the New England Journal of Medicine, researchers at UC San Francisco and UC Irvine show the device is as effective as a laboratory test and can detect cases even among people who have trouble producing phlegm samples from deep within their lungs.
“For patients who can’t produce sputum, like children or people with HIV, tongue swabs move the needle from ‘no diagnosis possible at this clinic’ to ‘accurate molecular testing here now,’” said Adithya Cattamanchi, MD, MPH, a professor of Medicine at UC Irvine, adjunct professor at UCSF, and co-lead author of the study.
The results have already led the World Health Organization to issue its first recommendation for this kind of TB test. The device, which operates on battery power, is so easy to use that the providers in the study learned how after reading instructions.

MiniDock MTB uses a technology, similar to the one used in hospital-based COVID tests, to detect the DNA of mycobacterium tuberculosis, the bacteria that cause TB.
Global health experts have long sought a more rapid form of this type of “point-of-care” testing, so patients do not need to wait days or even weeks to find out their tests results. Today’s “point-of-care” TB tests use technology dating back to the 1800s that reveal tuberculosis in sputum with a dye and microscope.
“Patients who today would have to leave a clinic undiagnosed could soon be diagnosed and treated the same day,” Cattamanchi said. “We hope to see more widespread adoption as evidence like ours stacks up.”
Authors: Other UCSF authors are Midori Kato-Maeda, MD; Tessa Mochizuki, MPH; Patrick P.J. Phillips, PhD; Payam Nahid, MD, MPH; and Adithya Cattamanchi, MD, MPH. For all authors, see the paper.
Funding: National Institutes of Health (U01AI152087, R01AI190419); U.S. State Department; European and Developing Countries Clinical Trials Partnership; German Federal Ministry of Research, Technology and Space; National Institute for Health and Care Research (UK); Department of Biotechnology, India; and the Gates Foundation.

